 The second was an acquisition offer from a European biotech. Since then AdAlta had been working toward meeting certain validation criteria and milestones. The first, a licensing agreement with a Big Pharma, had started in July 2008. "We found ourselves again in Australia with limited private venture capital groups investing and a need to raise further capital."Ī DEAL ISN’T A DEAL UNTIL THE MONEY IS IN YOUR BANK ACCOUNTĪt the time, AdAlta had two important deals in the works. It was 2009, and Cobb had been CEO of AdAlta for only 18 months. This outside interest spurred another round of angel investments that added another year of funding for the company. Serry invested as an angel investor, and his involvement eventually led to other high-net-worth (HNW) individuals as well as the BioAngels in SA (South Australia) joining this round of funding, which was enough for another year.įurther feedback from potential Big Pharma and biotech partners on possible immunogenicity risks with a foreign antibody-based technology pushed AdAlta’s focus to the i-body technology, the human version of the shark single-domain antibody. “I presented to all types of investors, including venture capital, but again, the technology was very early-stage, so there was little interest.” That changed when she met Leon Serry, the founder of the first ASX-listed biotech company, Circadian. But other partners were more interested in hearing about the shark antibody and i-body-based technologies that were far less-developed at the time.īased on this feedback and interest from Big Pharma, Cobb decided to pivot to the shark antibody-based technology. Feedback provided by Big Pharma and other potential partners was that the peptide half-life could be a long-term issue, and the company would need to demonstrate clinical efficacy. 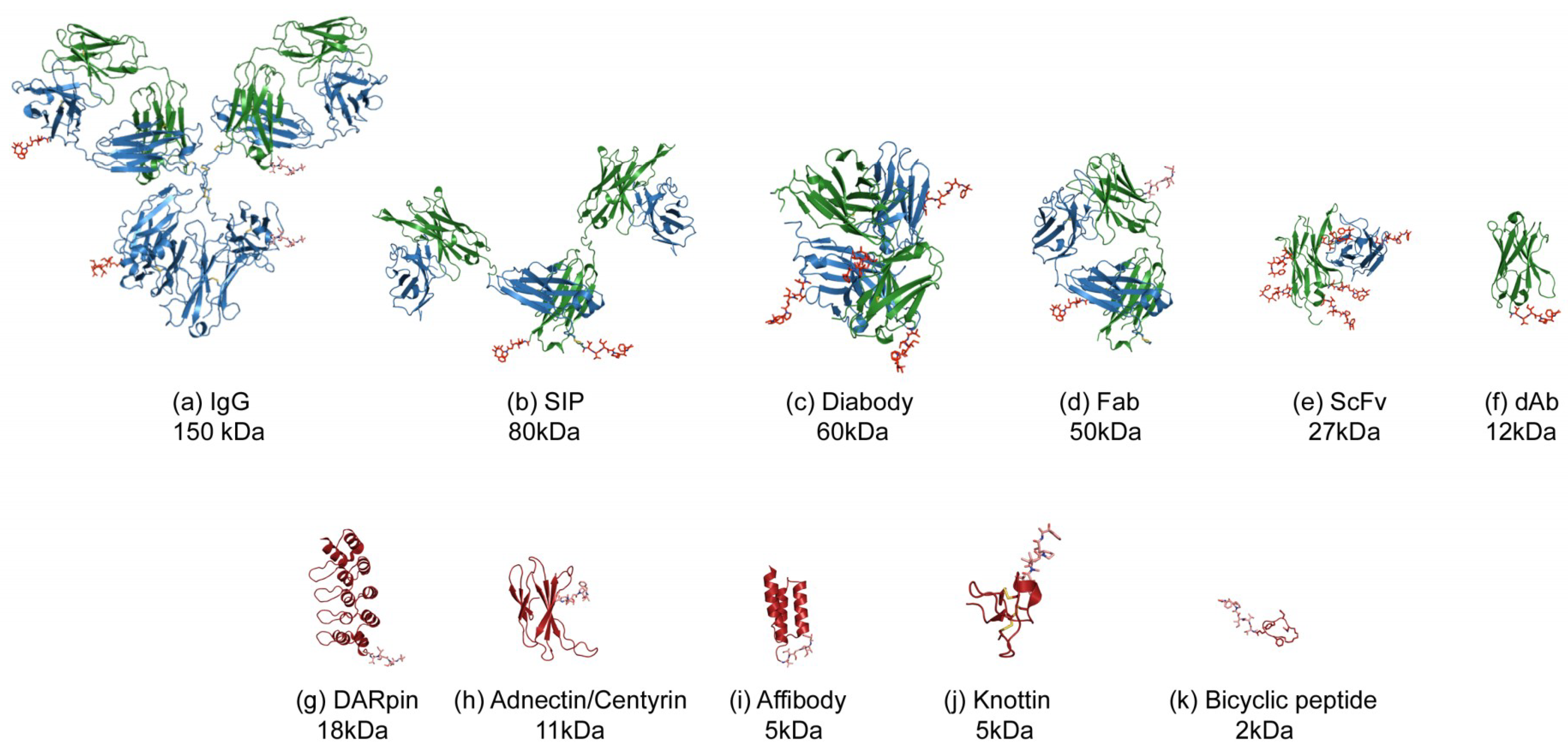
Initially, AdAlta focused on the technology that was more developed: the phage display library of peptides, with several lead candidates in the area of inflammation. "So significant work - more than our funding would support - still needed to be done." "For example, the i-body library had been designed but not validated and screened on one to two targets," Cobb explains. The CRC partners invested 12 months' worth of funding to get AdAlta started, but the technology (i.e., the libraries) was still very early in development and untested. a library of compounds that mimic the shape of shark single-domain antibodies, made with human protein as a scaffold (now called AdAlta's i-body library).a well-screened and developed peptide library.THE NEED TO PIVOT BASED ON CUSTOMER FEEDBACKĪdAlta started with three pieces of IP from an academic collaboration known as the Cooperative Research Centre for Diagnostics (the CRC), made up of multiple academic institutes and commercial parties in the field of developing novel diagnostic tests and technologies.Īll three pieces of IP were based on using phage display with the following novel libraries: and Australian biotech markets, led the company through the global financial crisis (GFC), and managed multiple iterations of the company’s lead candidate as it advances to the clinic. Since then, she says it’s been “a roller-coaster ride of personal growth” as she has navigated the vastly different terrains of the U.S. It’s now been 12 years since Cobb started the company, which develops treatments for fibrotic (i.e., scarring) diseases and is listed on the Australian Securities Exchange (ASX). “They never expected us to last past our first 12 months of funding.” That’s what Sam Cobb, managing director and CEO of Australian biotech AdAlta, recalls some of the original academic shareholders telling her years after the company had succeeded.  
By Dan Schell, Editorial Director, Life Science Leader 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
